Warning: Undefined array key "adf" in /www/wwwroot/petroleumrefine.com/wp-content/plugins/rejected-wp-keyword-link-rejected/wp_similarity.php on line 41
Warning: Undefined array key "sim_pages" in /www/wwwroot/petroleumrefine.com/wp-content/plugins/rejected-wp-keyword-link-rejected/wp_similarity.php on line 42
Both petrol and diesel are obtained from crude oil that has been squashed deep in the bowels of our planet. Both are equally ubiquitous: diesel powers larger engines, such as those of trucks, boats and cranes, while petrol powers smaller engines, such as those of cars and bikes. Ever wonder why that is the case? Also, what is it about diesel that makes it more efficient and suitable for steady motion, whereas petrol often causes shoulder-jolting acceleration?
Both fuels are equally ubiquitous because both fuels are equally important. Each has its own pros and cons. Understanding the difference between the fuels will reveal these various facets and the answers to the queries above.
Crude oil is a haphazard medley of hydrocarbons. These molecules of hydrogen and carbon form either long, linear chains or rings called cyclic compounds. The boiling point of a chain or ring is proportional to its size. This is obvious, as breaking a long, extensive chain of molecules is more strenuous than breaking a small or scanty chain of molecules. We can exploit this property to refine the oil or separate the chains from each other.
Fractional distillation
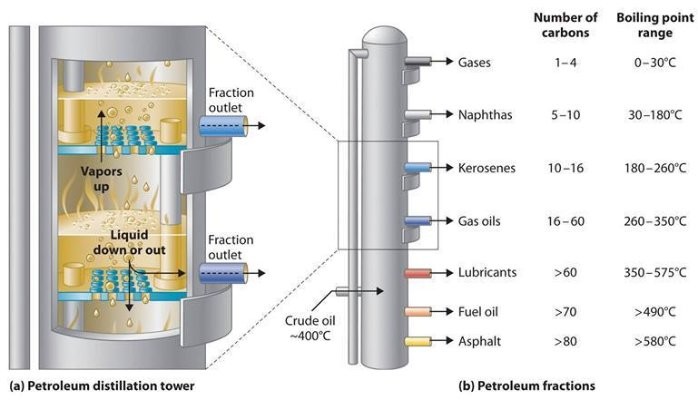
The extracted crude oil is first poured into a gigantic furnace. The furnace or column is then heated. The heating distills the crude oil, and this process is called fractional distillation because it, as the name suggests, causes the crude oil to fractionalize. Vertically, different regions of the furnace are subjected to different temperatures. The temperature, which is the lowest at its head, increases as we move towards its feet, where it is at a maximum.
Remember that longer chains are more difficult to break than shorter chains. Therefore, subjecting the different regions of the furnace to different temperatures allows us to extract carbon chains and rings of different lengths from the oil. Fractionalizing then allows us to extract various fuels from crude oil.
At the top of the furnace, lighter carbon chains or fuels exhibiting lower boiling points of 40-205℃ are obtained, such as LPG and petrol, while in the middle, denser fuels exhibiting higher boiling points of 250-350℃ are obtained, such as diesel. At the bottom of the barrel, the densest compounds are obtained, such as lubricants.
It is now obvious why petrol can cause shoulder-jolting acceleration: as a consequence of being much lighter, it ignites much more quickly than the dense diesel fuel does. However, as a consequence of being denser, diesel packs more hydrocarbons and therefore more energy per unit of volume than petrol does.
Typically, petrol comprises a recurring chain of no more than nine carbon atoms, while chains constituting diesel contain at least fourteen carbon atoms. Diesel cannot evaporate as rapidly as petrol, but being more glutinous (it is for its viscous, oily texture that it is often called diesel oil), it only packs 132 x 106 J per gallon, while petrol packs 155 x 106 J per gallon. Consequently, vehicles fueled by diesel are more mileage-oriented than vehicles fueled by petrol.