Warning: Undefined array key "adf" in /www/wwwroot/petroleumrefine.com/wp-content/plugins/rejected-wp-keyword-link-rejected/wp_similarity.php on line 41
Warning: Undefined array key "sim_pages" in /www/wwwroot/petroleumrefine.com/wp-content/plugins/rejected-wp-keyword-link-rejected/wp_similarity.php on line 42
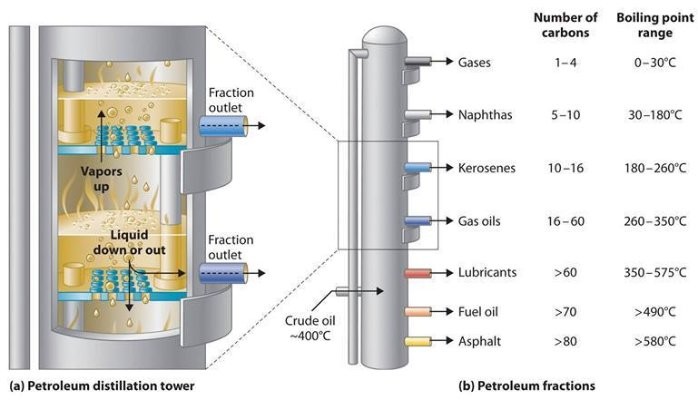
The captains of the oil industry were amongst essentially the most successful entrepreneurs of any century, reaping big income from oil, natural gas, and their byproducts and constructing business empires that soared to capitalism’s heights. Oil even grew to become a consider some of probably the most complex geopolitical struggles in the final quarter of the twentieth century, ones still playing out in the present day. Oil has touched all our lives in other ways as well. Reworked into petrochemicals, it is all around us, in just about every modern manufactured thing, from the clothes we put on and the medicines we take to the supplies that make up our computer systems, countertops, toothbrushes, operating shoes, automobile bumpers, grocery luggage, flooring tiles, and on and on and on. Certainly, the products from petrochemicals have performed as great a task in shaping the trendy world as gasoline and gas oils have in powering it. It appears at first a rooster-and-egg sort of query: Which came first—the gas pump or the automobile pulling as much as it? Gasoline was round earlier than the invention of the interior combustion engine but for many years was considered a ineffective byproduct of the refining of crude oil to make kerosene, an ordinary gas for lamps by way of much of the nineteenth century. Oil refining of the day—and into the first years of the twentieth century—relied on a comparatively simple distillation process that separated crude oil into parts, referred to as fractions, of different hydrocarbon compounds (molecules consisting of various arrangements of carbon and hydrogen atoms) with different boiling points. crude Oil Distillation Heavier kerosene, with more carbon atoms per molecule and a higher boiling point, was thus simply separated from lighter gasoline, with fewer atoms and a decrease boiling point, as well as from different hydrocarbon compounds and impurities within the crude oil combine. Kerosene was the keeper; gasoline and different compounds as well as natural gas that was usually found alongside oil deposits, have been usually just burned off.

Then in the primary 2 many years of the twentieth century horseless carriages in growing droves got here on the lookout for gas. Researchers had discovered early on that the internal combustion engine ran greatest on light fuels like gasoline but distillation refining just did not produce sufficient of it—only about 20 p.c gasoline from a given amount of crude petroleum. Whilst oil prospectors extended the range of productive wells from Pennsylvania via Indiana and into the huge oil fields of Oklahoma and Texas, the inherent inefficiency of the existing refining process was virtually threatening to carry again the automotive industry with gasoline shortages.
The issue was solved by a pair of chemical engineers at Commonplace Oil of Indiana—company vice president William Burton and Robert Humphreys, head of the lab at the Whiting refinery, the world’s largest at the time. Burton and Humphreys had tried and failed to extract extra gasoline from crude by adding chemical catalysts, but then Burton had an idea and directed Humphreys to add stress to the standard heating course of used in distillation. Under both heat and stress, it turned out that heavier molecules of kerosene, with up to sixteen carbon atoms per molecule, “cracked” into lighter molecules akin to those of gasoline, with four to 12 carbons per molecule, Thermal cracking, as the method got here to be referred to as, doubled the effectivity of refining, yielding 40 percent gasoline. Burton was issued a patent for the process in 1913, and shortly the pumps had been protecting tempo with the ever-rising automobile demand.
In the subsequent a long time other chemical engineers improved the refining process even additional. In the 1920s Charles Kettering and Thomas Midgley, who would later develop Freon (see Air Conditioning and Refrigeration), discovered that adding a form of lead to gasoline made it burn smoothly, stopping the unwanted detonations that brought about engine knocking. Tetraethyl lead was a regular ingredient of nearly all gasolines until the 1970s, when environmental issues led to the event of efficiently burning gasolines that did not require lead. One other major breakthrough was catalytic cracking, the challenge that had escaped Burton and Humphreys. Within the thirties a Frenchman named Eugene Houdry perfected a course of using sure silica and alumina-primarily based catalysts that produced even more gasoline via cracking and did not require excessive strain. In addition, catalytic cracking produced forms of gasoline that burned extra efficiently.