Crude oil, also called petroleum, is a complex mixture of carbon and hydrogen (hydrocarbons), which exist as a liquid in the earth’s crust. Crude oil has many compositions, some is black, thick and tar like, whereas different crude oils are lighter in coloration and thinner. The carbon and hydrogen in crude oil are thought to have originated from the stays of microscopic marine organisms that have been deposited at the bottom of seas and oceans and was transformed at excessive temperature and pressure into crude oil and natural gas.
This oil and gas migrates upward by way of the porous rock, as it’s much less dense than the water which fills the pores. The oil and gas is trapped by a layer of impermeable rock by way of which they can’t circulation. A number of various kinds of oil and gas “traps” exist; a standard dome formed by folded sedimentary rocks. Crude oil is obtained by drilling a gap into the reservoir rock (sandstone, limestone and so forth.) and pumping it out.
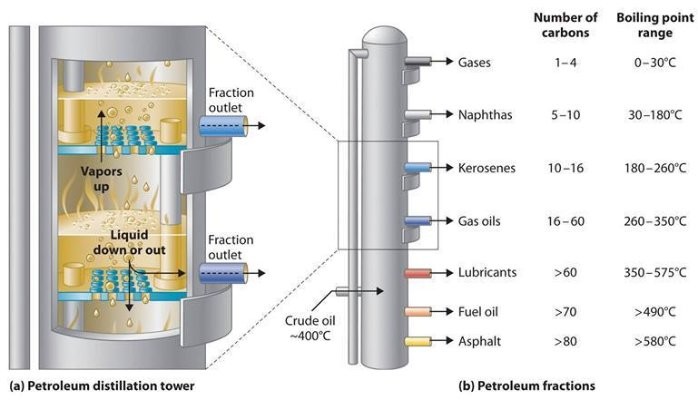
Petroleum refining is the means of separating the various compounds present in crude petroleum. This course of known as fractional distillation the place the crude oil is heated; the various of the compounds boil at completely different temperatures and change to gases; and are later recondensed again into liquids. Fossil Fuels
The principle which is used is that the longer the carbon chain, the higher the temperature at which the compounds will boil.
Boiling Points and Hydrocarbons
 The crude petroleum is heated and changed into a gas. The gases are passed by a distillation column which turns into cooler as the top will increase. See the figure on the left. When a compound in the gaseous state cools under its boiling point, it condenses into a liquid. The liquids could also be drawn off the distilling column at various heights.
The crude petroleum is heated and changed into a gas. The gases are passed by a distillation column which turns into cooler as the top will increase. See the figure on the left. When a compound in the gaseous state cools under its boiling point, it condenses into a liquid. The liquids could also be drawn off the distilling column at various heights.
Although all fractions of petroleum find uses, the greatest demand is for gasoline. petroleum equipment distributors One barrel of crude petroleum accommodates only 25-35% gasoline. Transportation calls for require that over 50% of the crude oil be transformed into gasoline. To meet this demand some petroleum fractions have to be converted to gasoline. This could also be finished by “cracking” – breaking down giant molecules of heavy heating oil; “reforming” – changing molecular constructions of low high quality gasoline molecules; or “polymerization” – forming longer molecules from smaller ones. Conversion Oil Refining
For instance if decane is heated to about 500 C the covalent carbon-carbon bonds begin to interrupt through the cracking process. Many kinds of compounds together with alkenes are made during the cracking process. Alkenes are formed because there aren’t sufficient hydrogens to saturate all bonding positions after the carbon-carbon bonds are broken.
If you have any queries with regards to where and how to use Refinery, you can contact us at our internet site.