Desalting
Crude oil often contains water, inorganic salts, suspended solids and water-soluble trace metals. The first step in the refining process is to remove these contaminants by desalting (dehydration) in order to reduce corrosion, plugging and fouling of equipment, and to prevent poisoning the catalysts in processing units. Chemical desalting, electrostatic separation and filtering are three typical methods of crude-oil desalting. In chemical desalting, water and chemical surfactants (demulsifiers) are added to the crude oil, heated so that salts and other impurities dissolve into the water or attach to the water, and are then held in a tank where they settle out. Electrical desalting applies high-voltage electrostatic charges in order to concentrate suspended water globules in the bottom portion of the settling tank. Surfactants are added only when the crude oil has a large amount of suspended solids. A third, less common process involves filtering heated crude oil using diatomaceous earth as a filtration medium.
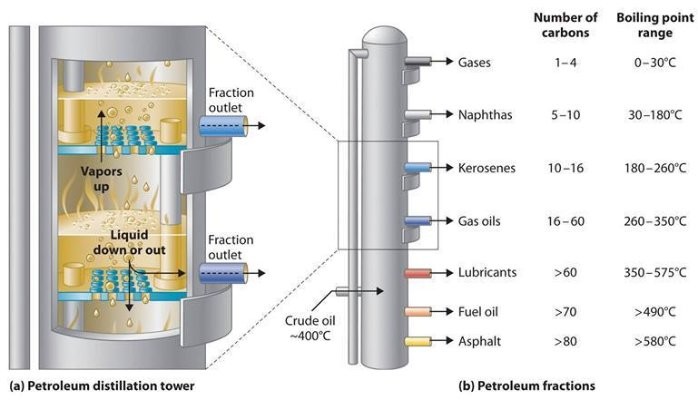
In chemical and electrostatic desalting, the crude feedstock is heated to between 66 °C and 177 °C, to reduce viscosity and surface tension for easier mixing and separation of the water. The temperature is limited by the vapour pressure of the crude-oil feedstock. Both methods of desalting are continuous. Caustic or acid may be added to adjust the pH of the water wash, and ammonia added to reduce corrosion. Waste water, together with contaminants, is discharged from the bottom of the settling tank to the waste water treatment facility. The desalted crude oil is continuously drawn from the top of the settling tanks and sent to an atmospheric crude distillation (fractionating) tower. (See figure 2.)
Figure 2. Desalting (pre-treatment) process
Inadequate desalting causes fouling of heater tubes and heat exchangers in all refinery process units, restricting product flow and heat transfer, and resulting in failures due to increased pressures and temperatures. Overpressuring the desalting unit will cause failure.
Corrosion, which occurs due to the presence of hydrogen sulphide, hydrogen chloride, naphthenic (organic) acids and other contaminants in the crude oil, also causes equipment failure. Corrosion occurs when neutralized salts (ammonium chlorides and sulphides) are moistened by condensed water. Because desalting is a closed process, there is little potential for exposure to crude oil or process chemicals, unless a leak or release occurs. A fire may occur as a result of a leak in the heaters, allowing a release of low-boiling-point components of crude oil.
There is the possibility of exposure to ammonia, dry chemical demulsifiers, caustics and/or acids during desalting. Where elevated operating temperatures are used when desalting sour crude oils, hydrogen sulphide will be present. Depending on the crude feedstock and the treatment chemicals used, the waste water will contain varying amounts of chlorides, sulphides, bicarbonates, ammonia, hydrocarbons, phenol and suspended solids. If diatomaceous earth is used in filtration, exposures should be minimized or controlled since diatomaceous earth can contain silica with a very fine particle size, making it a potential respiratory hazard.